 
Most part of the refiners do not consider the hydrotreating of petrochemical grade naphtha in their refining scheme due to the low contaminants content presented in the light straight run naphtha, for this reason, the hydrotreating alternative is highly effective but imposes a significant rise in the operating costs related to hydrogen availability and available processing capacity in existing hydrotreating units. 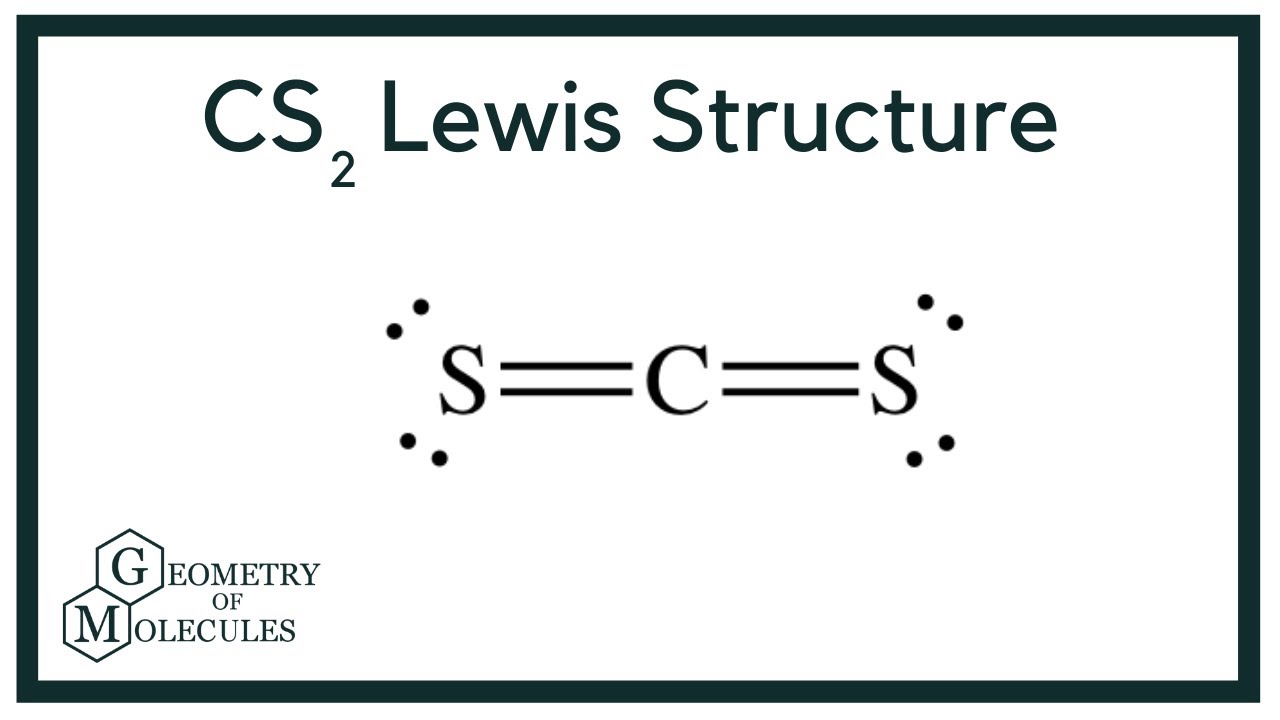
The H2S is recovered at the top of the stripping tower and treated in the sulfur recovery unit. In this case, the CS2 is converted into H2S according to the following reaction: A basic process flow diagram for a typical naphtha hydrotreating unit is presented in Figure 3.įigure 3 – Basic Process Flow Scheme for a Typical Naphtha Hydrotreating Unit The first option adopted by refiners to remove CS2 from naphtha is the hydrotreating units. The concentration of CS2 varies with the origin of the crude oils, according to the literature, the North American shale oil presents a higher concentration (50 to 250 ppm in mass) of this contaminant while some light crudes can present concentrations below 1,0 ppm in mass.Īvailable Routes to Deal with CS2 in Petrochemical Grade Naphtha Normally the origin of CS2 is related to the crude oil production process, in some cases, the producers apply triazine as an H2S scavenger, and the triazine degradation during the refining process produces Carbon Disulfide. The light fraction is collected in the top and commercialized as petrochemical grade naphtha.Īs aforementioned, the Carbon Disulfide tends to concentrated in the light fraction of naphtha. The straight run naphtha produced in the top of the atmospheric column is pumped to a naphtha split column where the heavy fraction is collected in the bottom and directed to the gasoline pool, in markets with high demand for middle distillates this stream can be added to diesel and jet fuel, according to the specification limits. Normally, the petrochemical grade naphtha is composed of the lighter fraction of straight run naphtha, as presented in Figure 2.įigure 2 – Basic Process Flow Diagram for a Typical Crude Oil Distillation Unit with Naphtha Splitter. 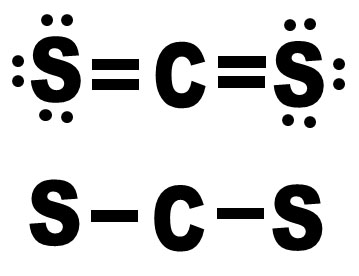
How is Produced the Petrochemical Naphtha in a Crude Oil Refinery? For these reasons, controls of the Carbon Disulfide in the petrochemical grade naphtha have been a great concern in the downstream industry in recent years. Due to his boiling point tends to concentrate in the lighter fraction of naphtha and their high chemical stability leads the CS2 to be stable to steam cracking process, reaching the most critical steps of petrochemical processes. The Carbon Disulfide is a potent poison to the catalysts applied to petrochemical processes (Ziegler-Natta) and can induce fragility and imperfections in the polymeric chain, mainly in the case of Isoprene which is an intermediate to rubber production. An interesting case is the content of Carbon Disulfide (CS2) in the petrochemical naphtha. Furthermore, the higher availability of lighter crude oils favors the oversupply of lighter derivatives that facilitate the production of petrochemicals against transportation fuels as well as the higher added value of petrochemicals in comparison with fuels.įigure 3 present an overview of the trend of growing the petrochemical market in short term.įigure 1 – Growing Trend in the Demand by Petrochemical Intermediates (Deloitte, 2019) - Note: Bars represent total demand (million metric tons or MMT), circles represent total capacity (MMT).Īs presented in Figure 1, is expected significant growth in the petrochemicals demand in short term and the refiners are looking for closer integration with petrochemical assets in order to improve their refining margins and ensure participation in a growing market, replacing the transportation fuels which presents lower margins and in contraction demand.ĭespite these advantages, as mentioned above, the petrochemicals maximization in the refining hardware presents some challenges mainly related to complying with the strict specifications of petrochemicals, once the crude oil refining processes are designed to produce derivatives with relatively low purity when compared with petrochemicals. 
New technologies like additive manufacturing (3D printing) has the potential to produce a great impact on the transportation demands, leading to even more impact over the transportation fuels demand. With the improvement in fuel efficiency, the growing market of electric vehicles tends to decline the participation of transportation fuels in the global crude oil demand. The growing demand for petrochemicals imposes great technologic challenges to refiners especially related to the stricter specifications of petrochemical derivatives in comparison with transportation fuels, requiring even more operational efficiency and optimization efforts from refiners. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
